The periodic table is a cornerstone of chemistry, organizing all known elements in a way that reveals patterns in their properties and behaviors. Understanding its structure is key to grasping fundamental chemical concepts. One of the most important distinctions within the periodic table is the division between metals and nonmetals. This article will focus on nonmetals, specifically addressing the question: Where Are Nonmetals Located On The Periodic Table?
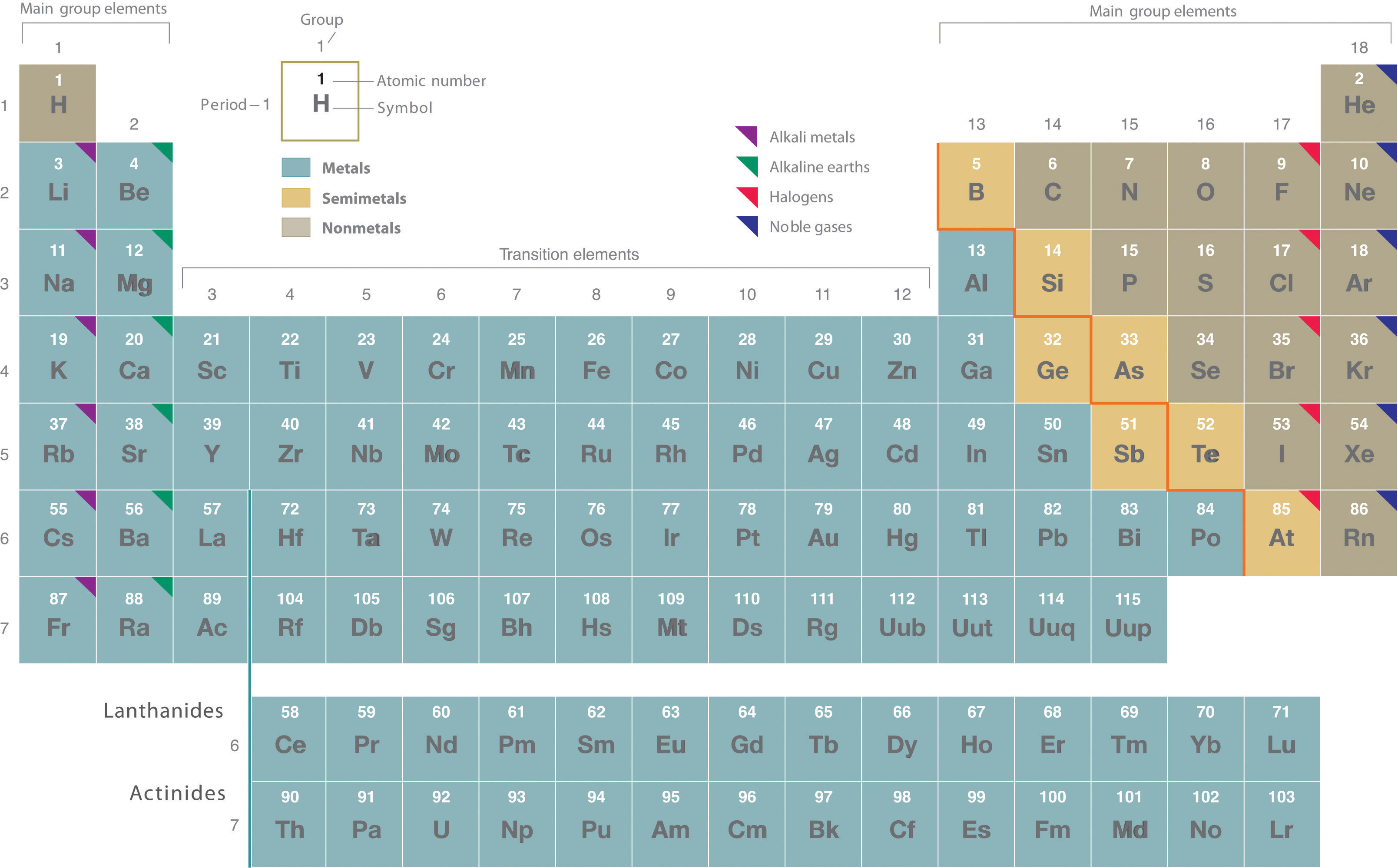
To understand the location of nonmetals, it’s essential to first grasp the basic layout of the periodic table. Elements are arranged in rows called periods (numbered 1 to 7) and columns known as groups (numbered 1 to 18). This arrangement isn’t arbitrary; it’s based on increasing atomic number (Z), which represents the number of protons in an atom’s nucleus. The number of protons defines the element’s identity and dictates its chemical properties.
 Periodic Table of the Elements
Periodic Table of the Elements
Identifying Nonmetals on the Periodic Table
Visually, the periodic table is often depicted with a bold, zigzag line that starts between Boron (B) and Aluminum (Al) and descends diagonally towards Polonium (Po) and Astatine (At). This line serves as a crucial guide for distinguishing between metals and nonmetals.
Nonmetals are predominantly located on the right-hand side and upper part of the periodic table, above and to the right of this diagonal line. In contrast, metals occupy the left-hand side and the bottom sections, below and to the left of the line. Elements that border this line, like silicon (Si) and germanium (Ge), are classified as semimetals or metalloids, exhibiting properties intermediate between metals and nonmetals.
Properties of Nonmetals
Nonmetals possess a set of characteristic properties that set them apart from metals. These include:
- Poor Conductors: Unlike metals, nonmetals are generally poor conductors of heat and electricity. This is due to the way their electrons are arranged and how they participate in bonding.
- Lack of Luster: Nonmetals typically do not have a shiny, metallic appearance. They are often dull and can come in various colors.
- States of Matter: Nonmetals can exist in all three states of matter at room temperature and pressure:
- Gases: Examples include oxygen (O), nitrogen (N), chlorine (Cl), and all the noble gases.
- Liquids: Bromine (Br) is a nonmetal that exists as a liquid at room temperature.
- Solids: Sulfur (S), phosphorus (P), carbon (C), and iodine (I) are solid nonmetals. Solid nonmetals are often brittle, meaning they break easily when hammered or stretched, unlike the malleable and ductile nature of metals.
Key Groups of Nonmetals
While most nonmetals are clustered on the right side of the periodic table, it’s helpful to consider specific groups that are exclusively or predominantly nonmetallic:
- Group 17: The Halogens: This group, located just before the noble gases, consists entirely of nonmetals: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). Halogens are known for their high reactivity and tendency to form salts when they react with metals.
- Group 18: The Noble Gases: Occupying the far right of the periodic table, Group 18 is composed of helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). These are all nonmetals and are famous for their inert or unreactive nature, existing as monatomic gases.
- Other Significant Nonmetals: Beyond these groups, other crucial nonmetals include hydrogen (H), which, although placed in Group 1, is a nonmetal. Oxygen (O), sulfur (S), selenium (Se) in Group 16, nitrogen (N), and phosphorus (P) in Group 15, and carbon (C) in Group 14 are also important nonmetals.
Semimetals: The Borderline Elements
It’s worth briefly mentioning semimetals (also known as metalloids) in the context of nonmetals. These elements—boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), tellurium (Te), and polonium (Po)—lie along the zigzag line and exhibit properties of both metals and nonmetals. Their intermediate nature makes them crucial in semiconductor technology.
Conclusion
In summary, nonmetals are found predominantly in the upper right portion of the periodic table, above and to the right of the zigzag line that divides metals from nonmetals. They are characterized by their poor conductivity of heat and electricity, lack of luster, and brittle nature when solid. Key groups of nonmetals include the halogens (Group 17) and the noble gases (Group 18). Understanding the location and properties of nonmetals is fundamental to comprehending chemical reactivity and the diverse world of elements. By using the periodic table as a map, we can easily predict and understand the basic classification of each element as a metal, nonmetal, or semimetal.


